Current and Emerging Treatments in Melanoma
Resectable Disease
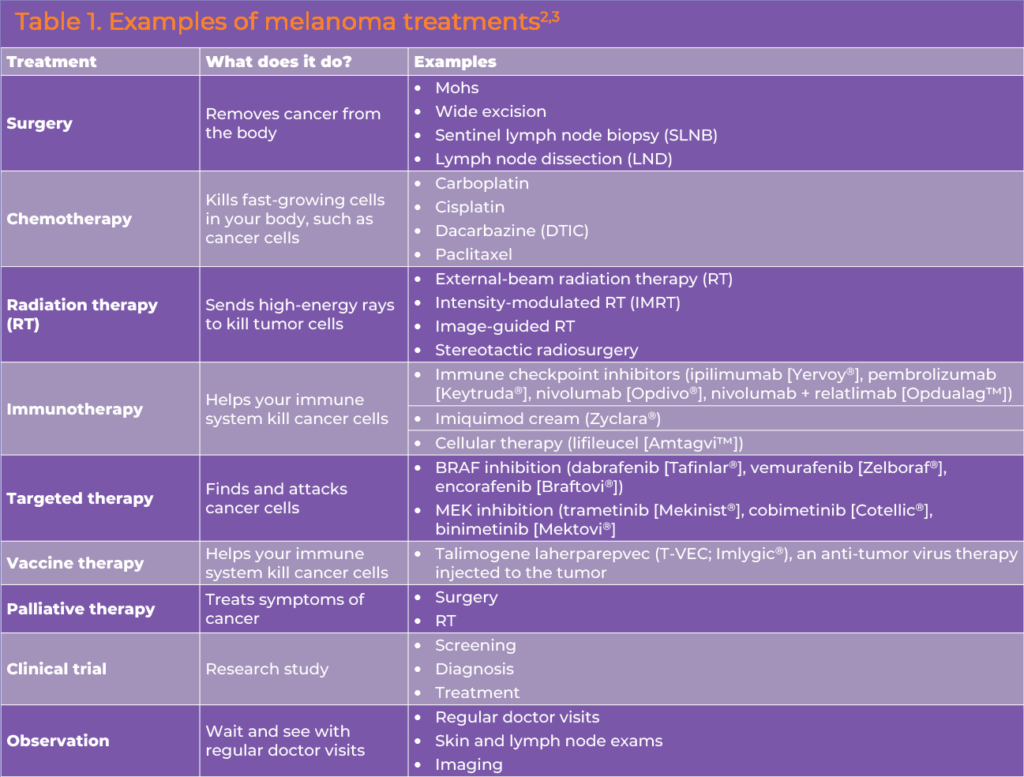
Local and systemic therapies are available for resectable melanoma. Treatment planning is largely dependent on the stage of disease (Table 1).
Surgery1
Excision
Surgical excision is the mainstay of therapy for localized melanoma.1 Surgical planning should aim for margins proportional to the microstage of the primary lesion.1 The 5-year survival rate for patients with localized cutaneous melanoma is >99%.2
Sentinel lymph node biopsy (SLNB)1
Lymphatic mapping and SLNB may be used to evaluate the presence of metastasis in the regional lymph nodes in cases that are clinically node negative with a primary tumor ≥ 0.8 mm thick and inform decision making regarding use of regional lymph node dissection (LND) and adjuvant therapy.1 Ideally, these procedures should be done at the time of wide excision of the primary lesion.
Complete LND1
Complete LND is considered to gain regional control in cases with microscopically- or macroscopically-positive SLN(s). For cases of micrometastatic melanoma, active surveillance using ultrasound of the draining nodal basin is commonly used instead of complete LND. A complete regional lymphadenectomy is an option for selected populations.
Systemic Therapy
Post-operative (Adjuvant) Therapy
Patients at high risk for recurrence after complete resection are eligible for adjuvant systemic therapy with an immune checkpoint inhibitor (ICI) or targeted therapy regimen.1 Key clinical trial data supporting these options are summarized in Table 2.
Adjuvant immune checkpoint inhibition
The NCCN Clinical Practice Guidelines for Melanoma include adjuvant monotherapy with an anti-programmed death-1 (PD-1) ICI, namely pembrolizumab or nivolumab, for patients with stage IIB/IIC melanoma.3
These agents have been shown to improve recurrence-free survival (RFS) for pathologic stage IIB and IIC melanoma.1 Assessment of overall survival (OS) outcomes require longer follow-up.1 However, the final 9-year results from the phase 3 CheckMate 238 study of adjuvant nivolumab versus ipilimumab did not show an OS benefit with nivolumab.4
Decisions on whether to use an adjuvant ICI should entail shared decision-making with eligible patients regarding associated benefits and risks in the context of factors such as age, performance status, personal/family history of autoimmune disease, absolute risk of recurrence, and risk of long-term immunotoxicities.3
Of note, the benefits of single-agent ICI adjuvant therapy were observed regardless of programmed death-ligand 1 (PD-L1) expression or BRAF mutation profile.1
Targeted therapy
For patients with a BRAF V600 mutation, adjuvant dabrafenib plus trametinib is an option based on results of the COMBI-AD study (Table 2).3
Perioperative Therapy
The National Comprehensive Cancer Network (NCCN) Clinical Practice Guideline for Melanoma includes several neoadjuvant systemic therapy options for certain patients with:
- clinically evident resectable stage III melanoma, with nodal basin disease, or isolated in-transit metastasis,
- extensive nodal metastases if upfront resection deemed too morbid,
- resectable oligometastatic stage IV disease, or
- clinically evident recurrent disease in the nodal basin following a formal nodal basin resection.3
Preferred regimens in the guidelines include nivolumab plus ipilimumab (category 1) and pembrolizumab (category 2a).3 Clinical trials supporting these options are summarized in Table 3.16-19 Other regimens include nivolumab with or without relatlimab and dabrafenib plus trametinib for BRAF V600-mutated melanoma (all category 2a).3 Neoadjuvant pembrolizumab did not significantly increase toxicity in the perioperative period compared with adjuvant therapy, and surgical outcomes were not negatively impacted. Overall survival data are not available yet. The United States Food and Drug Administration (FDA) has not approved these regimens, pending the completion of randomized phase 3 studies.
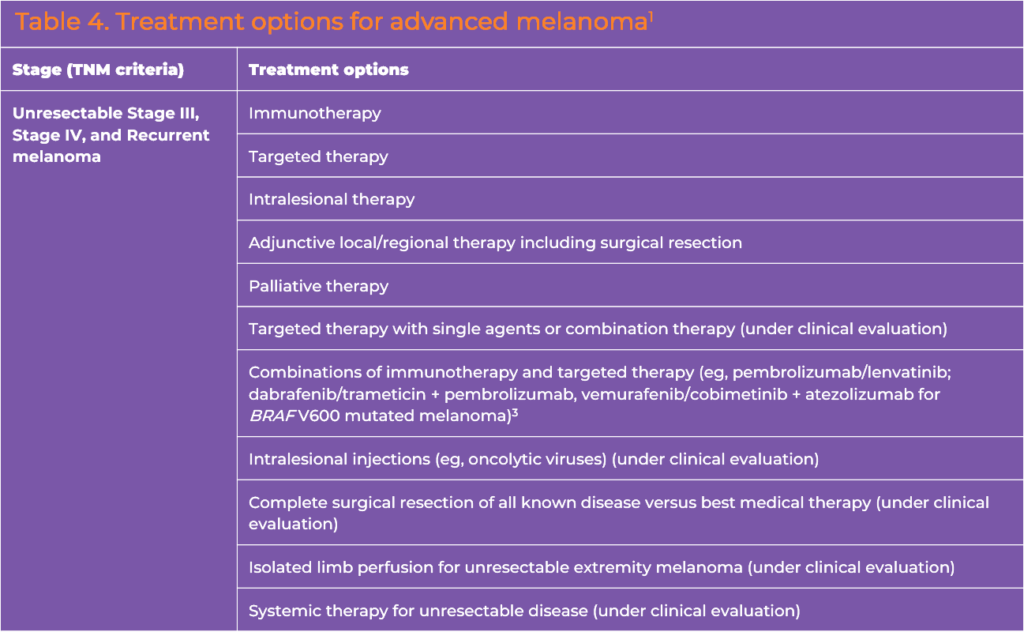
Unresectable Stage III-IV or Recurrent Disease
Systemic therapy is the primary option for patients with unresectable stage III-IV or recurrent disease (Table 4).3 Overall survival benefits have been demonstrated in randomized trials of ICIs and of BRAF/MEK targeted therapies in this setting.
Preferred regimens in the NCCN melanoma guidelines are nivolumab/ipilimumab (category 1), nivolumab/relatlimab (category 1) or ICI monotherapy with pembrolizumab or nivolumab (category 1).3 Of note, combination immune checkpoint blockade is the preferred first-line approach. Other recommended first-line regimens include BRAF/MEK combination targeted therapy (dabrafenib /trametinib, vemurafenib/cobimetinib, encorafenib/binimetinib) for BRAF V600 mutation positive melanoma (all category 1).3 Pembrolizumab with low-dose ipilimumab is another option included in the guidelines as a category 2b recommendation.
Immune checkpoint inhibitors
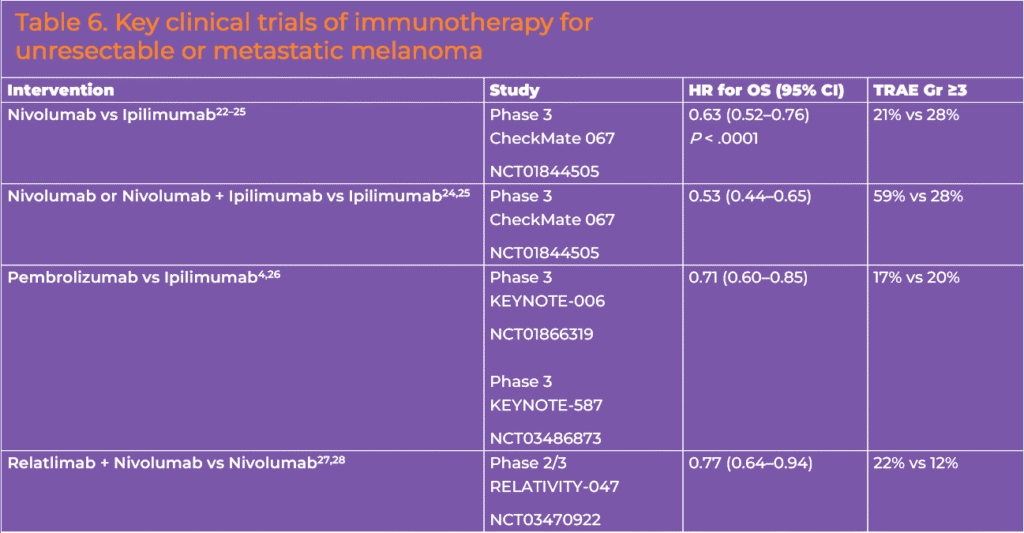
Several ICIs are approved by the FDA for the treatment of advanced (unresectable or metastatic) melanoma, including PD-1 inhibitors nivolumab and ipilimumab, the cytotoxic T-lymphocyte antigen 4 (CTLA-4) inhibitor ipilimumab, and the lymphocyte activation gene 3 (LAG-3) inhibitor relatlimab (the latter is approved in a fixed-dose formulation with nivolumab). Each has demonstrated clinical activity and a survival benefit in randomized clinical trials (Table 6).
Multiple phase 3 trials are in progress to evaluate novel immunotherapeutics and combinations and to determine the optimal sequencing of immunotherapies (see Reading and Resources tab). A notable emerging anti-PD-1 and LAG-3 combination regimen—cemiplimab plus fianlimab—has also shown a PFS benefit as first-line therapy in patients who had not received a prior anti-PD-1 or anti-PD-L1 therapy.20 A phase I study of the combination demonstrated an objective response rate (ORR) of 63% and a median PFS of 15.3 months in the systemic therapy-naïve cohort.20 The median PFS was 13.3 months in the overall study population. This combination is being evaluated for melanoma in several phase 3 clinical trials (See Reading and Resources section). Based on studies of nivolumab and relatlimab and cemiplimab plus fianlimab, anti-PD-1/LAG-3 antibody combinations seem to be associated with lower overall and high-grade adverse events and discontinuations, compared to nivolumab plus ipilimumab.21-23
Targeted therapy
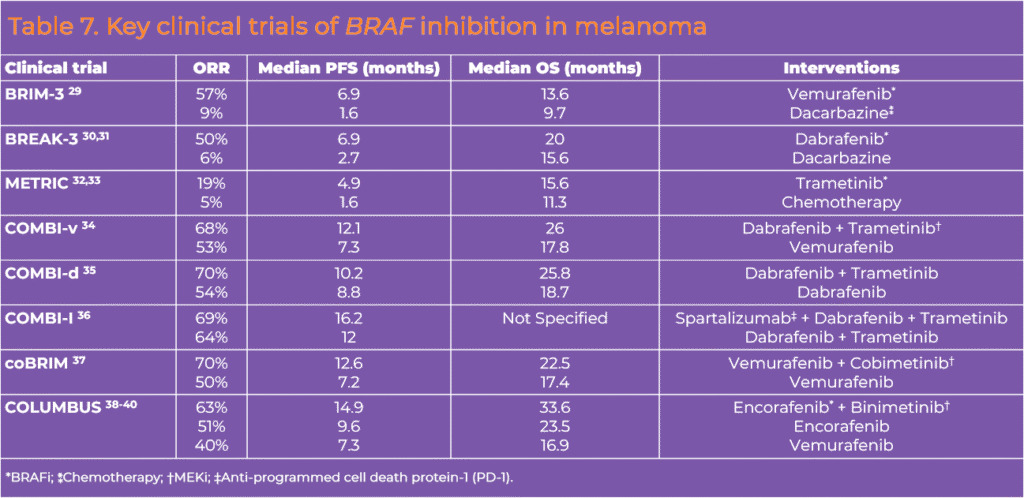
BRAF inhibition used in combination with MEK inhibition has been shown to improve PFS and OS compared with single-agent BRAF inhibition.
References
- National Cancer Institute. Melanoma Treatment (PDQ®) – Health Professional Version. Updated May 2, 2025. https://www.cancer.gov/types/skin/hp/melanoma-treatment-pdq
- American Cancer Society. Survival Rates for Melanoma Skin Cancer. Revised January 13, 2026. https://www.cancer.org/cancer/types/melanoma-skin-cancer/detection-diagnosis-staging/survival-rates-for-melanoma-skin-cancer-by-stage.html
- National Comprehensive Cancer Network®. NCCN Clinical Practice Guidelines in Oncology. Melanoma: Cutaneous. Version 1.2026. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1492
- Ascierto PA, Del Vecchio M, Merelli B, et al. Nivolumab for resected stage III or IV melanoma at 9 Years. N Engl J Med. 2026;394:333-342. https://doi.org/10.1056/NEJMoa2504966
- Long GV, Hauschild A, Santinami M, et al. Adjuvant dabrafenib plus trametinib in stage III BRAF-mutated melanoma. N Engl J Med. 2017;377:1813-1823. https://doi.org/10.1056/NEJMoa1708539
- Dummer R, Hauschild A, Santinami M, et al. Five-year analysis of adjuvant dabrafenib plus trametinib in stage III melanoma. N Engl J Med. 2020;383:1139-1148. https://doi.org/10.1056/NEJMoa2005493
- Larkin J, Del Vecchio M, Mandala M, et al. Adjuvant nivolumab versus ipilimumab (CheckMate 238 trial): Reassessment of 4-year efficacy outcomes in patients with stage III melanoma per AJCC-8 staging criteria. Eur J Cancer. 2022;173:285-296. https://www.sciencedirect.com/science/article/pii/S0959804922003926
- Eggermont AMM, Blank CU, Mandala M, et al. Adjuvant pembrolizumab versus placebo in resected stage III melanoma. N Engl J Med. 2018;378:1789-1801. https://doi.org/10.1056/NEJMoa1802357
- Eggermont AMM, Blank CU, Mandala M, et al. Longer follow-up confirms recurrence-free survival benefit of adjuvant pembrolizumab in high-risk stage III melanoma: Updated results from the EORTC 1325-MG/KEYNOTE-054 trial. J Clin Oncol. 2020;38:3925-3936. https://doi.org/10.1200/JCO.20.02110
- Eggermont AMM, Kicinski M, Blank CU, et al. Five-year analysis of adjuvant pembrolizumab or placebo in stage III melanoma. NEJM Evid. 2022;1:EVIDoa2200214. https://doi.org/10.1056/EVIDoa2200214
- Long GV, Luke JJ, Khattak MA, et al. Pembrolizumab versus placebo as adjuvant therapy in resected stage IIB or IIC melanoma (KEYNOTE-716): Distant metastasis-free survival results of a multicentre, double-blind, randomised, phase 3 trial. Lancet Oncol. 2022;23:1378-1388. https://doi.org/10.1016/S1470-2045(22)00559-9
- Luke JJ, Ascierto PA, Khattak MA, et al. Pembrolizumab versus placebo as adjuvant therapy in resected stage IIB or IIC melanoma: Final analysis of distant metastasis-free survival in the phase III KEYNOTE-716 study. J Clin Oncol. 2024;42:1619-1624. https://doi.org/10.1200/JCO.23.02355
- Luke JJ, Rutkowski P, Queirolo P, et al. Pembrolizumab versus placebo as adjuvant therapy in completely resected stage IIB or IIC melanoma (KEYNOTE-716): A randomised, double-blind, phase 3 trial. Lancet. 2022;399:1718-1729. https://doi.org/10.1016/S0140-6736(22)00562-1
- Kirkwood JM, Del Vecchio M, Weber J, et al. Adjuvant nivolumab in resected stage IIB/C melanoma: Primary results from the randomized, phase 3 CheckMate 76K trial. Nat Med. 2023;29:2835-2843. https://doi.org/10.1038/s41591-023-02583-2
- Amaral T, Ottaviano M, Arance A, et al. Cutaneous melanoma: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2025;36:10-30. https://doi.org/10.1016/j.annonc.2024.11.006
- Blank CU, Lucas MW, Scolyer RA, et al. Neoadjuvant nivolumab and ipilimumab in resectable stage III melanoma. N Engl J Med. 2024;391:1696-1708. https://doi.org/10.1056/NEJMoa2402604
- Patel SP, Othus M, Chen Y, et al. Neoadjuvant-adjuvant or adjuvant-only pembrolizumab in advanced melanoma. N Engl J Med. 2023;388:813-823. https://doi.org/10.1056/NEJMoa2211437
- Burton EM, Milton DR, Tetzlaff MT, et al. Long-term survival and biomarker analysis evaluating neoadjuvant plus adjuvant relatlimab (anti-LAG3) and nivolumab (anti-PD1) in patients with resectable melanoma. J Clin Oncol. 2025;43:2856-2862. https://doi.org/10.1200/JCO-25-00494
- Amaria RN, Postow M, Burton EM, et al. Neoadjuvant relatlimab and nivolumab in resectable melanoma. Nature. 2022;611:155-160. https://doi.org/10.1038/s41586-022-05368-8
- Hamid O, Lewis KD, Weise A, et al. Phase I study of fianlimab, a human lymphocyte activation gene-3 (LAG-3) monoclonal antibody, in combination with cemiplimab in advanced melanoma. J Clin Oncol. 2024;42:2928-2938. https://doi.org/10.1200/JCO.23.02172
- Dimitriou F, Hauschild A, Mehnert JM, Long GV. Double trouble: Immunotherapy doublets in melanoma-approved and novel combinations to optimize treatment in advanced melanoma. Am Soc Clin Oncol Educ Book. 2022;42:1-22. https://doi.org/10.1200/EDBK_351123
- Long GV, Carlino MS, McNeil C, et al. Pembrolizumab versus ipilimumab for advanced melanoma: 10-year follow-up of the phase III KEYNOTE-006 study. Ann Oncol. 2024;35:1191-1199. https://doi.org/10.1016/j.annonc.2024.08.2330
- Wolchok JD, Chiarion-Sileni V, Rutkowski P, et al. Final, 10-year outcomes with nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2025;392:11-22. https://doi.org/10.1056/NEJMoa2407417
- Wolchok JD, Chiarion-Sileni V, Gonzalez R, et al. Long-term outcomes with nivolumab plus ipilimumab or nivolumab alone versus ipilimumab in patients with advanced melanoma. J Clin Oncol. 2022;40:127-137. https://doi.org/10.1200/JCO.21.02229
- Wolchok JD, Chiarion-Sileni V, Gonzalez R, et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2017;377:1345-1356. https://doi.org/10.1056/NEJMoa1709684
- Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: Final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet. 2017;390:1853-1862. https://doi.org/10.1016/S0140-6736(17)31601-X
- Tawbi HA, Schadendorf D, Lipson EJ, et al. Relatlimab and nivolumab versus nivolumab in untreated advanced melanoma. N Engl J Med. 2022;386:24-34. https://doi.org/10.1056/NEJMoa2109970
- Tawbi HA, Hodi FS, Lipson EJ, et al. Three-year overall survival with nivolumab plus relatlimab in advanced melanoma from RELATIVITY-047. J Clin Oncol. 2025;43:1546-1552. https://doi.org/10.1200/JCO.24.01124
All URLs accessed March 23, 2026